Abstract
Introduction and aims:
Chimeric Antigen Receptor T-cell therapy (CAR-T) cell therapy has emerged as an effective therapy for relapsed haematological malignancies. Due to the unique nature of its mechanism of action and on-target, off-tumour toxicity, leading to myelosuppression and prolonged B-lymphocyte aplasia, the risk of infectious complications is substantially increased during the first year (12-55%) based on the data from the registration trials.1,2 Here we present real world data from a single UK centre on late infections in CAR-T treated patients.
Methods:
Retrospective data was collected on 40 consecutive patients treated with CAR-T cell therapy for any indication at Bristol Haematology and Oncology Centre between February 2019 and September 2021.
Results:
A total of 40 patients received Axicabtagene Ciloleucel (n=26), Tisagenlecleucel (n=12), Brexucabtagene Autoleucel (n=1) and JNJ-68284528 (CARTITUDE-1) (n=1) with a median follow-up duration of 24 months (range 11-42) months. Median age was 59 (range 16-76), 67.5% were male (M=27, F=13) and 80% received bridging therapy.
Median number of prior therapies received was 3 (range 2-5) with 40% of patients (n=16) having had a prior stem cell transplant. Of the total Non-Hodgkin lymphoma (NHL) patients (n=34), half (n=17) had progressive disease (PD) at the time of receiving CAR-T therapy while one-third of B-Acute lymphoblastic leukaemia (B-ALL) patients (n=4) had PD. 15% of patients (n=6) had 7 episodes of infections in the 30 days prior to CAR-T therapy and 10% (n=4) had moderate- severe pre-existing neutropenia.
All patients received anti-viral and anti-fungal prophylaxis with Acyclovir and Fluconazole according to institutional protocol. Pneumocystis Jiroveci prophylaxis with Co-trimoxazole or Pentamidine was started from day 28 of CAR-T. All patient received standard lymphodepletion with Fludarabine and Cyclophosphamide.
Significant neutropenia was observed in majority of patients (95%) with 7.5% having persisting neutropenia beyond 12 months. Of these patients, G-CSF therapy was used in 81% of patients (n=31). Hypogammaglobulinemia was present in 60% of patients following treatment (n=24) of which 37.5% (n=9) received at least one dose of Intravenous immunoglobulin (IVIg). 80% of patients developed Cytokine release syndrome and 32.5% had immune effector cell-associated neurotoxicity syndrome of any grade, with only 20% of patients (n=8) receiving steroid therapy.
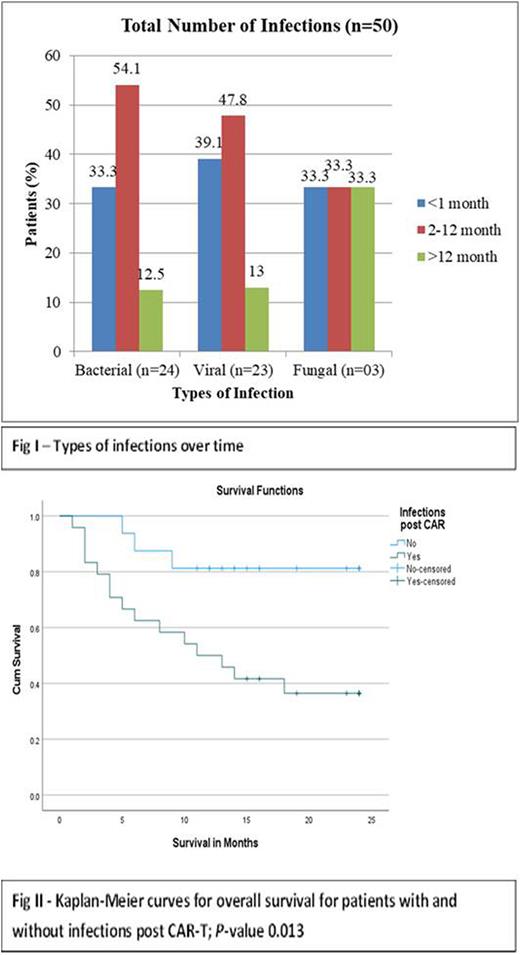
Total number of infections observed over the median follow-up period were 50, seen in 60% of patients (n=24). Types of infections over time are shown in Fig I. Highest number of infections (52%) were present in 2 - 12 month period (n=26) while 14% of all infections were observed after 12 months (n = 7). 42% of infections were mild (not requiring intervention), 46% moderate (requiring in-patient or out-patient treatment), 8% severe (requiring intensive care therapy) and 4% were fatal. In multivariate analysis, performance status was the only factor associated with higher infection risk (Hazard Ratio 2.83, P-value 0.027).
All patients with B-ALL (n=6) achieved CR at day 30 post CAR-T while 61% of NHL patients achieved Complete response.
With a median follow-up time of 24 months, 18 patients have died giving an overall survival of 55% for the entire cohort. The median overall survival of patients without any infections (A) was not reached while for patients with any infection (B) was 11 months (95% CI 3.8 - 18.2). The 2-year OS rates were 81.3% and 37.5% for Group A and B, respectively with p-value of 0.013 (Figure II).
Conclusion:
A small number of infections (mild-moderate) continued to occur after 12 months in our cohort of CAR-T patients suggesting that longer prophylaxis may be required in some patients on individualised basis.
Despite the shortcomings of this study including retrospective nature and small sample size, there is suggestion of adverse outcomes in patients developing any infections following CAR-T, necessitating aggressive prophylactic approach including use of IVIg.
References:
1. Schuster SJ, Bishop MR, Tam CS, et al. Tisagenlecleucel in adult relapsed or refractory diffuse large B-cell lymphoma. N Engl J Med 2019; 380: 45-56.
2. Abramson JS, Palomba ML, Gordon LI, et al. Lisocabtagene maraleucel for patients with relapsed or refractory large B-cell lymphomas (TRANSCEND NHL 001): a multicentre seamless design study. Lancet 2020; 396:839-852.
Disclosures
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal